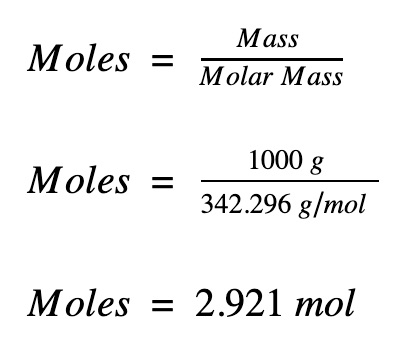Calculate the number of moles the following:52 g of He (finding mole from mass)12.044times 10^ {23} number of He atoms (finding mole from number of particles)

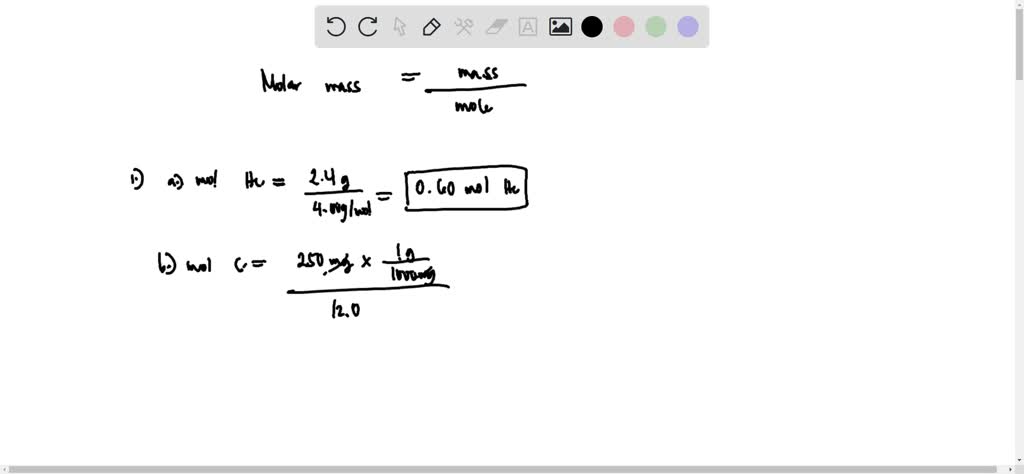
Calculate the number of moles in the following:(i) 28 g of He(ii) 46 g of Na(iii) 60 g of Ca(Atomic mass of He = 4 g, Na = 23 g, Ca = 40 g) - m92pdahgg

Calculate the number of moles of gas present in the container of volume 10 L at 300 K. If the manometer containing glycerin shows 5 m difference in level as shown diagram.