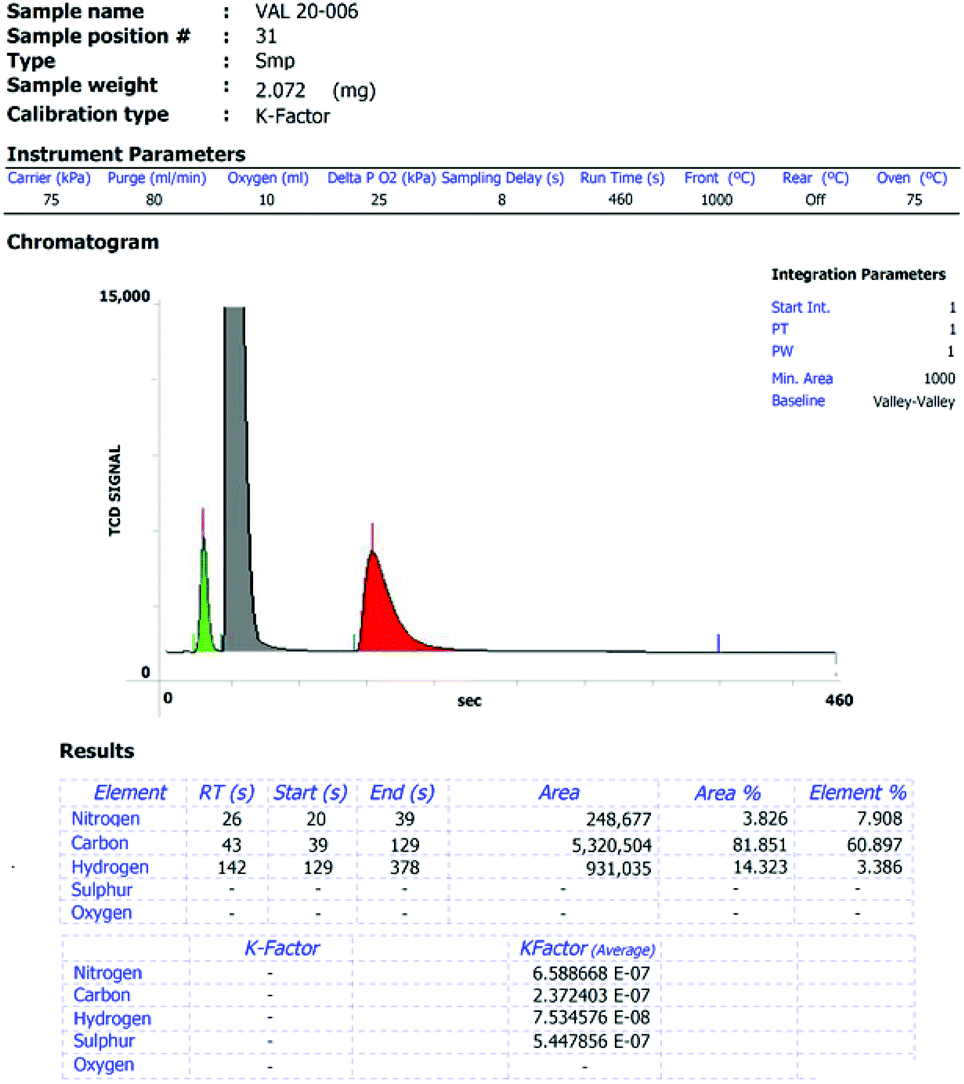
Identification and Characterization of an Isolated Impurity Fraction: Analysis of an Unknown Degradant Found in Quetiapine Fumarate | Waters
Elemental analysis: an important purity control but prone to manipulations - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QI01379C
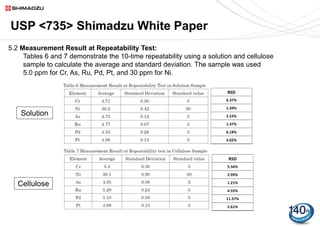
USP 735 as an Alternative to USP 233 for Elemental Impurity Analysis in Pharmaceutical Products | PPT

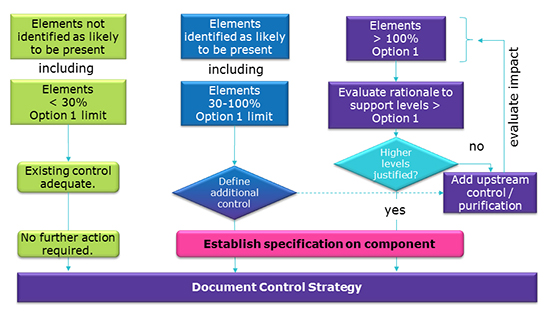
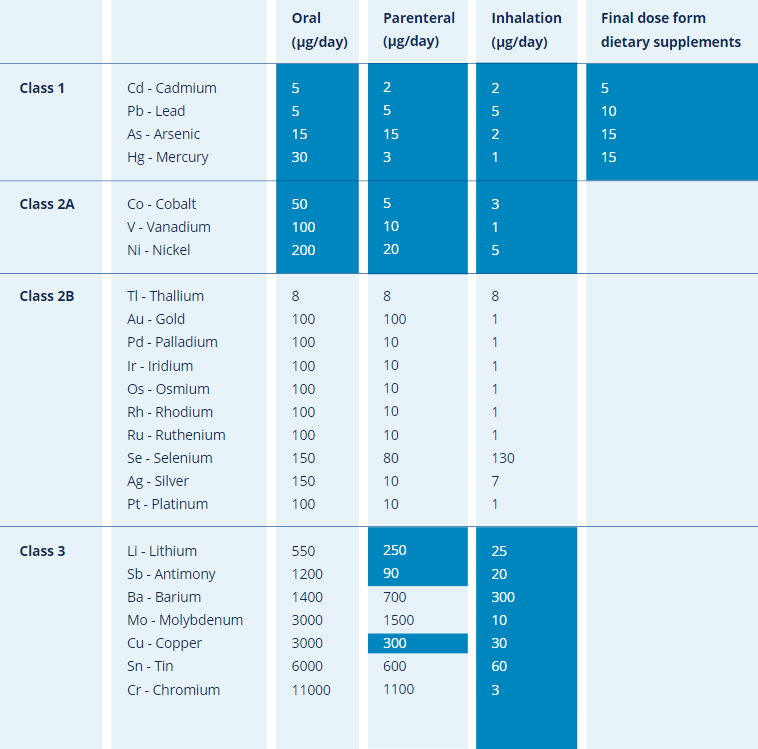
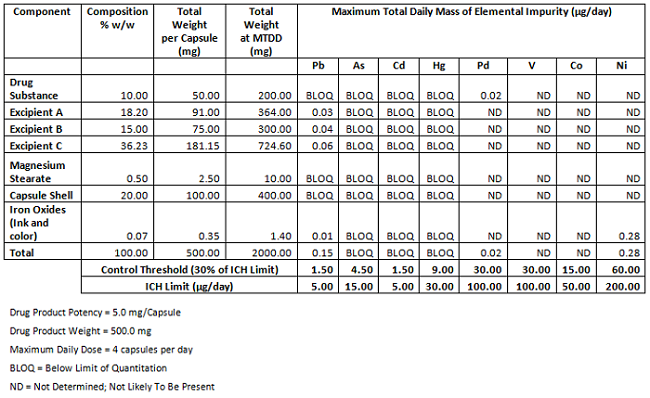
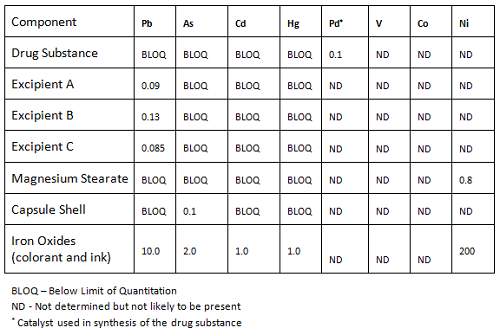
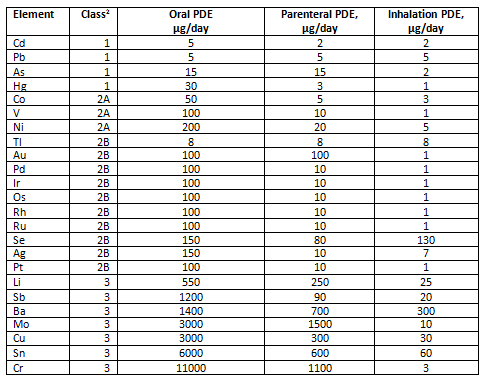
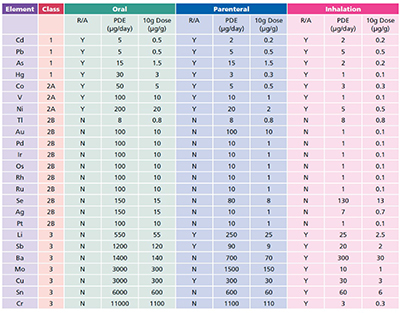
Calculation elemental impurities by Option 2b as per ICH Q3D. | How to calculate elemental impurities by Option 2b as per ICH Q3D (Guideline for Elemental Impurities)? | By Pharma Content | Facebook