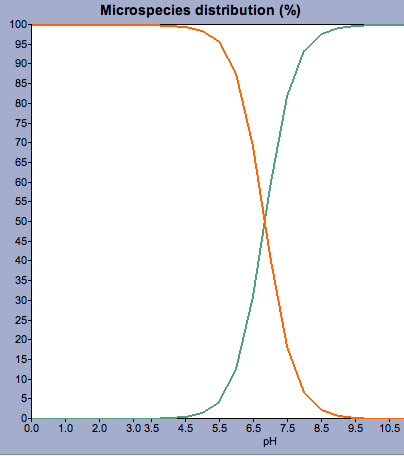
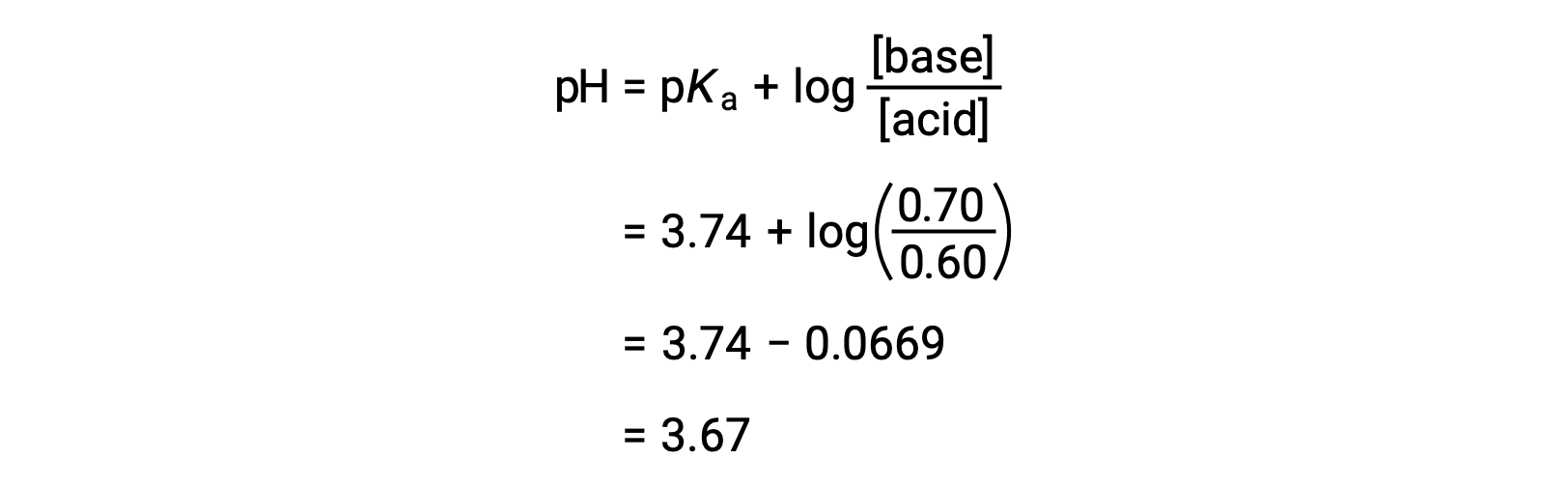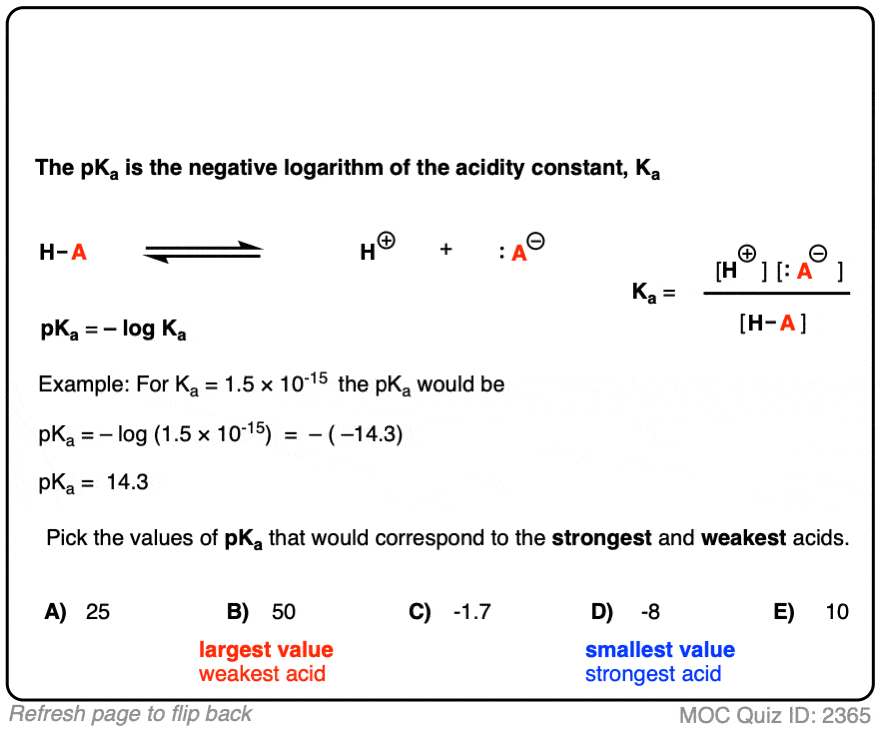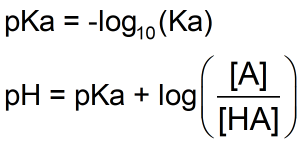SOLVED: Calculate the total solubility of the basic drug € at pH 6.0. Additional information for drug C: SB (intrinsic solubility) = 0.8 mg/mL, pKa = 8.2. St = S × (1 + [

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate.

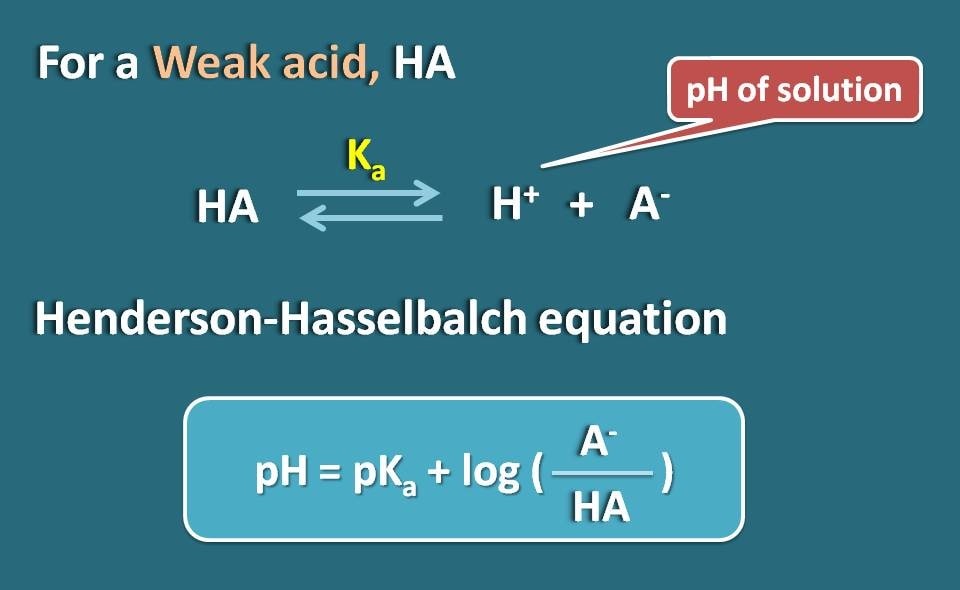
SOLVED: pH = pKa + log10 Using the Henderson-Hasselbalch equation as seen above, solve for pKa. Given the mixture of 0.30 M sodium acetate and 0.20 M acetic acid, what is the

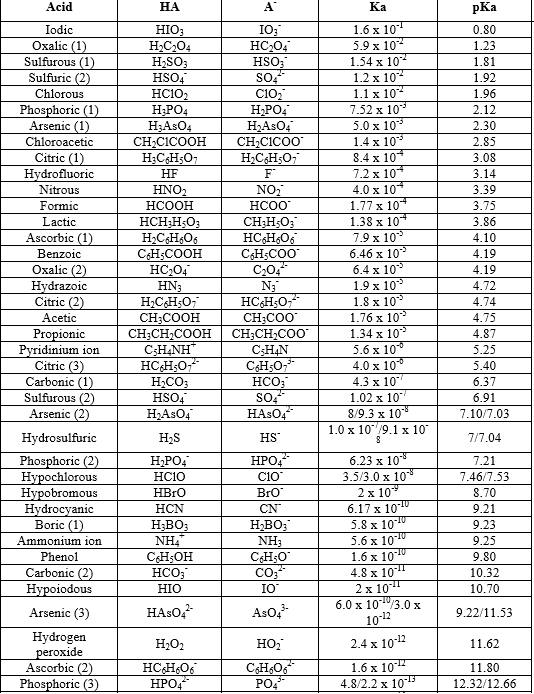
How should the acid dissociation constant pKa be measured? | Automatic Potentiometric Titrators | Faq | Kyoto Electronics Manufacturing Co.,Ltd.("KEM")