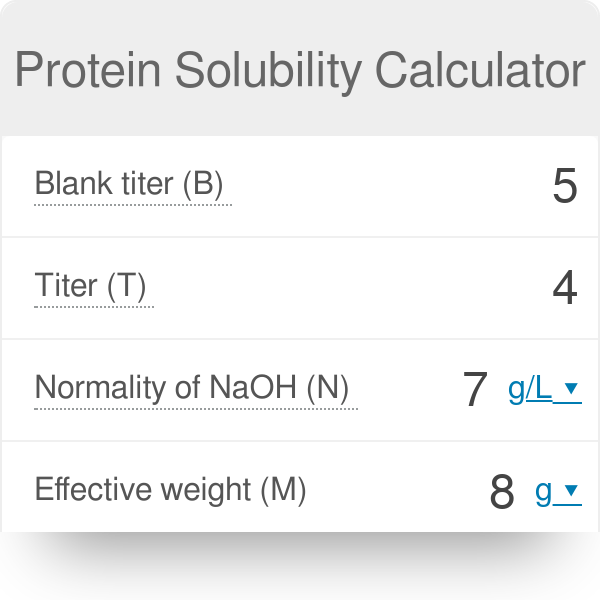
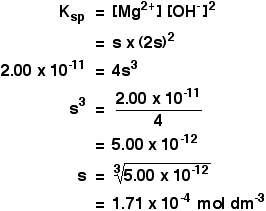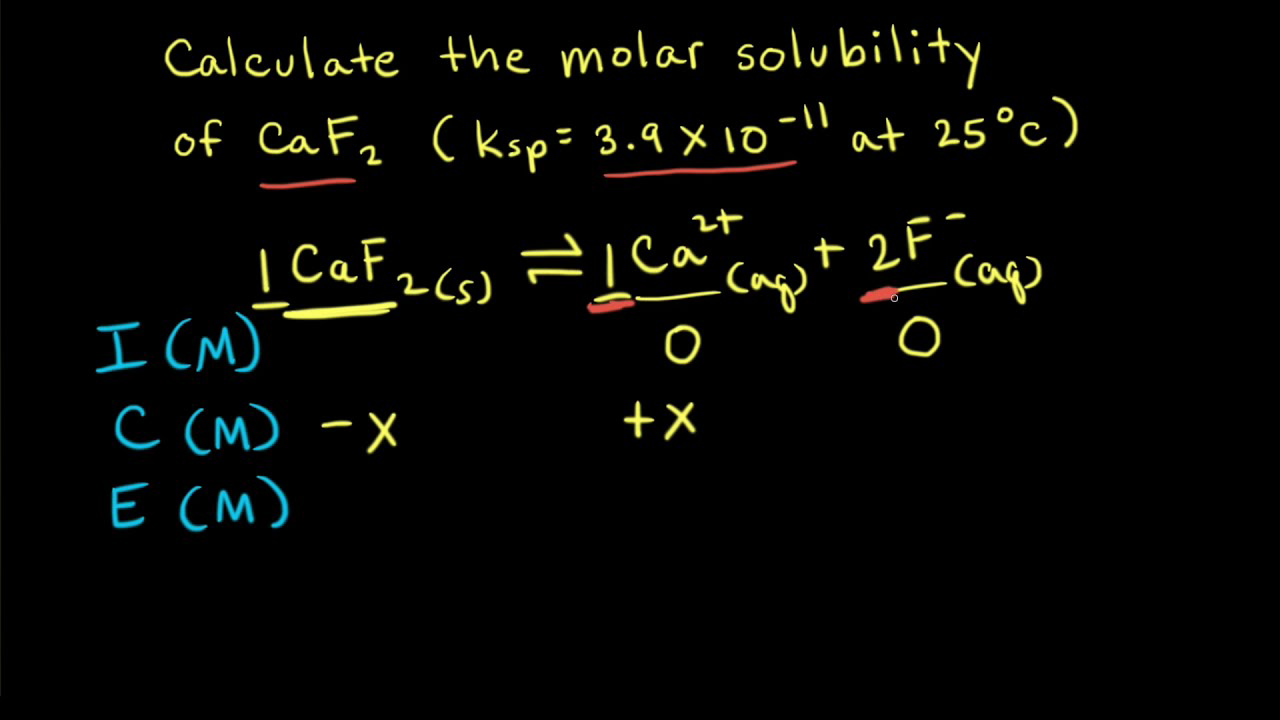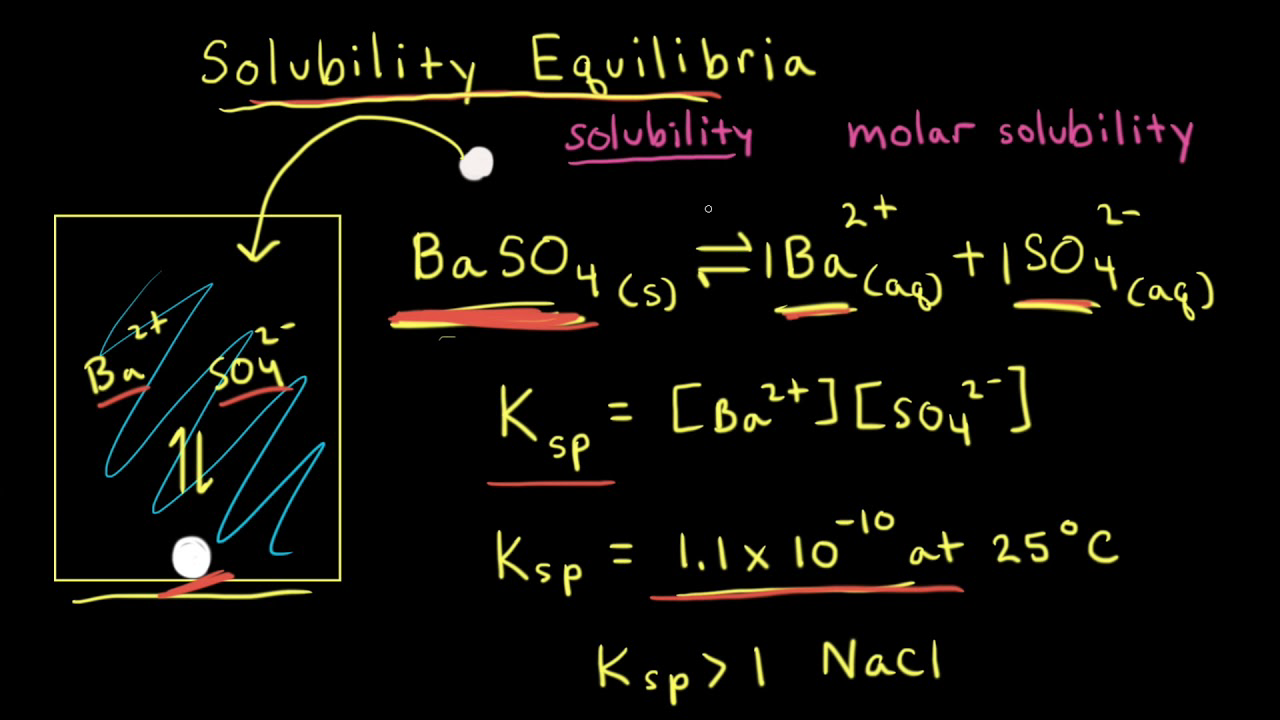✓ Solved: Calculate the % relative error in solubility by using concentrations instead of activities...

SOLVED: Calculate the total solubility of the basic drug € at pH 6.0. Additional information for drug C: SB (intrinsic solubility) = 0.8 mg/mL, pKa = 8.2. St = S × (1 + [